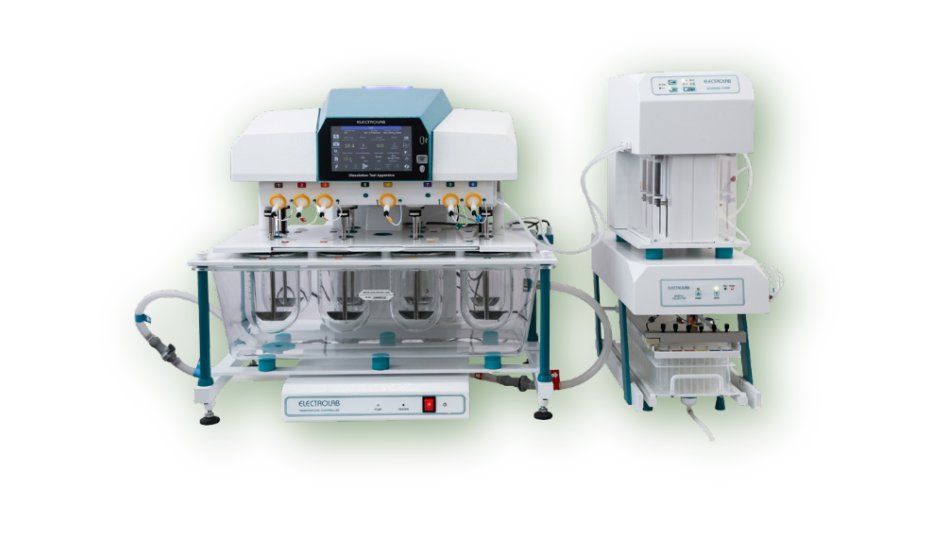
Electrolab Dissolution
Electrolab is India's leading manufacturer of dissolution testing equipment, with systems deployed across thousands of pharmaceutical quality control laboratories worldwide. The TDT and EDS series dissolution testers fully comply with USP, BP, EP, and IP pharmacopoeia requirements for basket and paddle apparatus.
MLYN Technologies provides expert service, calibration, and IQ/OQ/PQ qualification for Electrolab dissolution systems. Our team ensures your dissolution apparatus meets all pharmacopoeia mechanical calibration requirements and generates compliant, audit-ready test results.
Key Features
Technical Specifications
| Application | Tablet, Capsule, Transdermal Release |
| Apparatus Type | USP Type I (Basket), Type II (Paddle) |
| Vessel Volume | 900 mL / 1000 mL / 200 mL |
| Temperature Range | 25°C to 45°C ± 0.05°C |
| RPM Range | 25–250 RPM ± 1% |
| Vessel Configuration | 6 or 8 Vessels |
| Compliance | USP <711>, BP, EP, IP |
| Software | EDS Software with 21 CFR |